Mission Statement
The DCI Network accelerates solutions to complex healthcare problems by creating new multi-stakeholder alliances and consortia, and creating strategic roadmaps to implement solutions that scale.
Why?
Many of today’s challenges, such as real-world evidence, distributed clinical trials, multi-institutional data Linking, and new privacy standards, will require public-private collaborations. The pandemic has shown that improvements to health care cannot move quickly without coordinated efforts. Innovations that can’t be deployed at scale are a loss to the health and business sectors. We aim to drive efficiencies in the R&D process and healthcare delivery through more integrated data sharing and multistakeholder collaborations.
Members of DCI Network
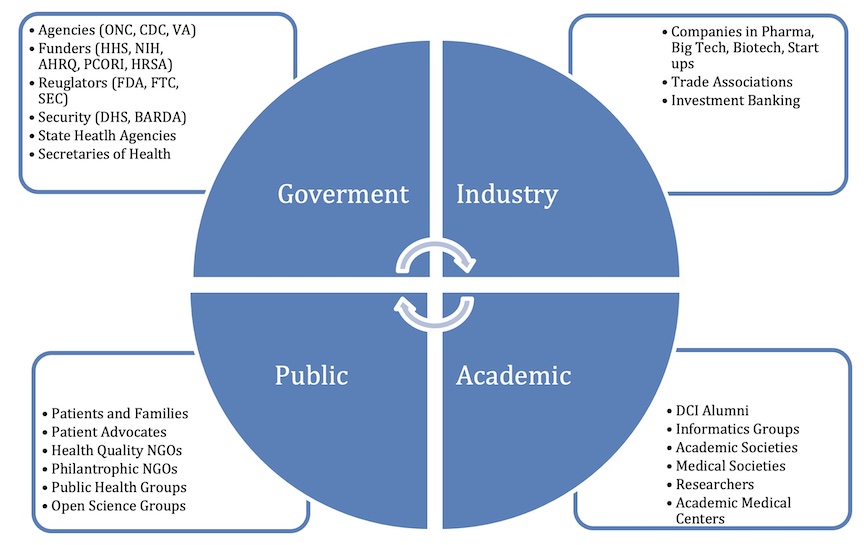
Our members are leaders from major biopharmaceutical companies, high-tech companies, healthcare providers, academic centers, and non-profit groups. Members are C-Suite leaders (CEO, CIO, CMIO) from non-profits, the private sector, academics, government, patients, and advocates dedicated to collaborative approaches to medicine and science.
CONVENER: The Division of Clinical Informatics (DCI) at Beth Israel Deaconess Medical Center (BIDMC) is a leading center for developing scalable models, informatics research, and policy-making since 1970.
Learn about our membership levels and join the Network today.
For more information, contact Dr. Yuri Quintana at yquintan@bidmc.harvard.edu
How?
The DCI Network brings together top thought leaders and decision-makers from the private sector, government, non-profits, and academia to discuss the most complex problems that require multistakeholder collaboration. A public webinar series will invite keynote speakers and host open discussions with public stakeholders. DCI Network convenes private meetings exclusively for DCI Network members to foster alliances that span the ecosystem. A steering committee of the DCI anchor network members would decide on the topics to focus on. We will create working groups to create roadmaps for solutions that can be implemented at scale and strategies for external funding. Activities aim to enhance, not duplicate, efforts of existing organizations. Membership will be by invitation only.
Stakeholder Benefit/Impact
-
DCI Network provides a safe harbor environment for collaboration of the private sector.
-
Alliances created by the DCI can accelerate innovation development and implementation at scale.
-
Create extensive and improved database models for both commercial and research initiatives.
-
White papers that could influence regulatory and funding policy.
-
Roadmaps will be used for strategies for how to implement solutions at-scale
Sample Topic Areas
-
Real World Evidence
-
New frameworks for accelerating approvals using Real-World Evidence.
-
Approaches for rapid collection of data on emerging pathogens and pandemics.
-
Strategies to accelerate recruitment into decentralized clinical trials.
-
Best data governance models for private and public stakeholders.
-
Precision Medicine
-
Approaches for validating and certifying AI-based systems.
-
New digital standards for medical labels and drug-drug interaction alerts.
-
Patient-Reported Outcomes
-
Best strategies for engaging and aggregating data directly from patients.
-
Strategies for using data from smart homes and remote monitoring.
-
Regulatory policy changes that enable better digital health data sharing.
Deliverables
-
Convene public webinars and events to identify the most urgent needs and opportunities.
-
Convene invitation-only events for DCI Network members.
-
Develop roadmaps for implementing solutions at scale via consortia.
-
Disseminate results from the DCI Network via white papers, webinars, and conferences.
-
Host networking events for DCI Network members to share thought leadership perspectives.
-
Propose strategies for turning roadmaps into funded projects with public and private funding.
-
Enhancing work of existing standards organizations by providing strategies for improved adoption.
Working Groups
We will identify stakeholders needed, detailed use cases, implementation and governance models, and funding strategies. We will produce high-level public white paper highlights for further public discussion. Roadmaps will be available to the public to help solve some of the country’s biggest challenges.
For 2023 we will be focusing on these areas:
- Improving Real World Evidence
- New frameworks for accelerating approvals using Real-World Evidence.
- Approaches for rapid collection of data on emerging pathogens and pandemics.
- Strategies to accelerate recruitment into decentralized clinical trials.
- Best data governance models for private and public stakeholders.
- Approaches for validating and certifying AI-based systems.
- New digital standards for medical labels and drug-drug interaction alerts.
- Improving Distributed Clinical Trials
- Improving how we recruit under-represented minorities to clinical trials.
- Develop strategies for retaining participants in trials
- Improve how we collect data from patients remotely and distribute it across sites.
- Improving Patient-Engagement
- Identifying the best approach for engaging patients is to enhance behavior change patients.
- Innovative strategies for collecting data from smart homes and remote monitoring.
- Ethics and standards data collection from wearables and social media for research
- Regulatory policy changes that enable better digital health data sharing across sites and sectors.
Proposed Working Groups for 2023
- Creating a precision oncology model that will aggregate clinical records, genomic, imaging, patient-reported data, and social determinants of health to support longitudinal life science research and the clinical decision for treating complex cancers requiring combination therapies. (Initial pilot with prostate cancer and hard-to-treat leukemia)
- Creating an approach to improve recruitment and retention of underrepresented monitories using data-intensive multi-site strategies to evaluate different approaches and serve as a data source for future trials
- Develop a framework for ethical data collection for longitudinal studies for interventions aimed at changing health behavior in patients at risk of chronic diseases (obesity, diabetes, cardiovascular, mental health)
To request to join one of these groups, please contact Yuri Quintana, Ph.D. at yquintan@bidmc.harvard.edu
Leadership and Convener
DCI Network is led by Yuri Quintana, Ph.D., the division chief of the Division of Clinical Informatics at the Beth Israel Deaconess Medical Center, a Harvard-affiliated teaching hospital. He created a global cancer disease registry and a global online community of practice. He leads implementations of the Alicanto platform used by Harvard-affiliated hospitals for large-scale communities of practice and performance improvement. He also leads the InfoSAGE initiative, a patient-centric platform and mobile app for medication and symptom management.
The Harvard-based Division of Clinical Informatics (DCI) at Beth Israel Deaconess Medical Center (BIDMC) is a leading center for developing scalable models, informatics research, and policy-making since 1970. DCI was among the first to create medical records, telemedicine, and patient-centric health technologies. DCI has implemented solutions worldwide and hosted international conferences on the secondary use of health data and health policy, which influenced the final version of GDPR. DCI is developing new technologies for the interoperability of national medication databases and innovative new mobile apps for collecting patient-reported outcomes for distributed clinical trials. For over 50 years, DCI has successfully convened multistakeholder groups to solve complex informatics and policy challenges and remains a trusted third-party convenor across industries and sectors.
Join: Visit the website https://www.dcinetwork.org to learn about our membership levels and join the Network today. For more information, contact Dr. Yuri Quintana at yquintan@bidmc.harvard.edu.











